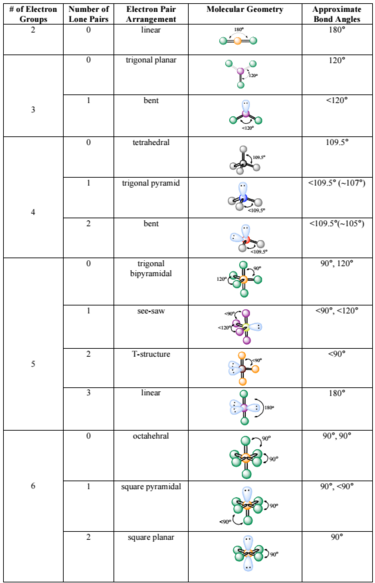
How To Calculate Bond Angle In Molecules
Calculate the moment of inertia of water molecule around the axis defined by the bisector of HOH bond. It is a neutralization by atom approach and neutralizes atoms with a 1 or -1 charge by removing or adding hydrogen where possible.

What Is The Bond Angle Of Of2 Study Com
The bond angles for molecules having a tetrahedral geometry are generally 1095 degrees but as there are double bonds it might be close to this angle but not precisely 1095 degrees.

How to calculate bond angle in molecules. The bond angles in BrF 5 are 90. DFT grid size on point group. The atom symbols for the atoms making an angle.
DFT grid on bond length. Bond length and bond order are two parameters that are associated with covalent bonds. The oxidation state of any chemically bonded carbon may be assigned by adding -1 for each bond to more electropositive atom H Na Ca B and 1 for each bond to more electronegative atom O Cl N P and 0 for each carbon atom bonded directly to the carbon of interest.
The covalent bond formed between two atoms in molecules whose electronegative difference exists is known as a polar covalent bond. But we cant neglect the effect of lone pair on a bond angle while calculating the molecular shape of the molecule. Bond bond energy kJ mol 1 CN in HCN 890 NN 994 CO 1078 Although each compound contains the same number of electrons and a strong triple bond in its molecule CO and HCN are.
For diatomic molecules like H 2 Cl 2 O 2 N 2 HCl HBr HI the bond enthalpies are equal to their dissociation enthalpy. TEQ The equilibrium bond angle for the above angle in degrees. The Lewis Model of Chemical Bonding.
BrF5 Molecular Geometry and Shape. BOND ANGLE ca-c3-c1 64784 110735 Calculated with empirical approach c1-c1-cx 56400 177990 same as c1-c1-c3 c1-cx-hc 48300 109750 same as c1-c3-hc c1-cx-cx 64200 111590 same as c1-c3-c3 DIHE IMPROPER ca-ca-ca-ha 11 1800 20 General improper torsional angle 2 general atom types. Hence it attracts more electrons and repulsion between the bond of nitrogen increased.
Same bondangle many molecules. How does molecule shape change with different numbers of bonds and electron pairs. Starting with bonded 12 bond length 13 bond angle and 14 dihedral angle interactions TraPPE relies on experimental data or electronic structure calculations to provide equilibrium bond lengths and angles the corresponding force constants and the dihedral potentials for new models.
Then compare the model to real molecules. Each molecule contains a strong triple bond with the following bond energies. In a π bond the regions of orbital overlap lie on opposite sides of the internuclear axis.
Here Ɵ bond angle. Products of moments of inertia. SO42- Shape As predicted by the VSEPR theory a molecule that consists of the central atom forming bonds with four atoms has a tetrahedral shape.
The angle formed by a water molecule is known to be 1045 and the bond moment of the O-H bond is -15D. A molecular vibration is a periodic motion of the atoms of a molecule relative to each other such that the center of mass of the molecule remains unchanged. All right lets move over to this carbon right here so this carbon has a triple-bond on the right side of it and so the fast way of doing this is if it has a triple-bond it must be SP hybridized here so SP hybridized and therefore the geometry would be linear with a bond angle of 180 degrees.
Dipole moment represents the bond moment it helps to calculate percentage ionic character of a covalent bond. In the case of polyatomic molecules bond enthalpies are usually the average values because the dissociation energy varies with each type of bond. Experimental and computed quantum mechanics thermochemical data for a selected set of 2069 gas-phase atoms and small molecules.
Core correlation - bond length. D With two nuclei around the central atom and one lone pair of electrons the molecular geometry of SnCl 2 is bent like SO 2 but with a ClSnCl bond angle of 95. Then the angle between the atoms is used to find the net dipole moment.
This neutralize_atoms algorithm is adapted from Noel OBoyles nocharge code. The dipole moment is calculated by multiplying the distance between the hydrogen and oxygen atoms by the difference in their charge. BrF5 Bond Angles.
Along the axis itself there is a node that is a plane with no probability of finding an electron. Covalent Bonding in H2 H. The constituent atoms repel each other in accordance with the VSEPR theory.
As there are two lone pairs present on the sulfur central atom in the H2S molecule hence it will contract the bond pair and this makes its shape appears like a bent structure. Bond order is the number of chemical bonds between two atoms and bond length is the distance between two nuclei of atoms that are covalently bonded together. Bond angle HOH 1045o and bond length OH 957.
Lets consider the Lewis structure for CCl 4. The most convenient way is. Bond angle or dihedral.
A pi bond π bond is a type of covalent bond that results from the side-by-side overlap of two p orbitals as illustrated in Figure 4. To determine the molecular geometry for Bromine Pentafluoride we go back to its Lewis structure. Find out by adding single double or triple bonds and lone pairs to the central atom.
Hence repulsion between the bond of nitrogen decreased and it makes NF3 bond angle lower. Two atoms constitutes a covalent bond. Because the lone pair of electrons occupies more space than the bonding pairs we expect a decrease in the ClSnCl bond angle due to increased LPBP repulsions.
Tools for comparing experimental and computational ideal-gas thermochemical properties. TK The harmonic force constants for the angle ITT-JTT- KTT in units of kcalmolrad2 radians are the traditional unit for angle parameters in force fields. We can draw the Lewis structure on a sheet of paper.
For trigonal pyramidal geometry the bond angle is slightly less than 1095 degrees around 107 degrees. Electrons in molecules are often delocalized between two or more atoms. Disulfide bonds in proteins are formed between the thiol groups of cysteine residues by the process of oxidative foldingThe other sulfur-containing amino acid methionine cannot form disulfide bondsA disulfide bond is typically denoted by hyphenating the abbreviations for cysteine eg when referring to ribonuclease A the Cys26Cys84 disulfide bond or the 2684 disulfide bond or.
Hence it causes the bond angle of NH3 higher than NF3. This article explains how to calculate bond order and bond length in an atomic level. Greater is the bond dissociation enthalpy greater is the bond strength.
For bent molecular geometry when the electron-pair geometry is tetrahedral the bond angle is around 105 degrees. In the case of NH3 nitrogen is more electronegative than hydrogen. From the Lewis structure it can be observed that Bromine has an expanded octet.
The SMARTS pattern checks for a hydrogen in 1 charged atoms and checks for no neighbors with a negative charge for 1 atoms and no neighbors with a positive charge for -1 atoms. Explore molecule shapes by building molecules in 3D. The typical vibrational frequencies range from less than 10 13 Hz to approximately 10 14 Hz corresponding to wavenumbers of approximately 300 to 3000 cm 1 and wavelengths of approximately 30 to 3 µm.
A sulfate ion.
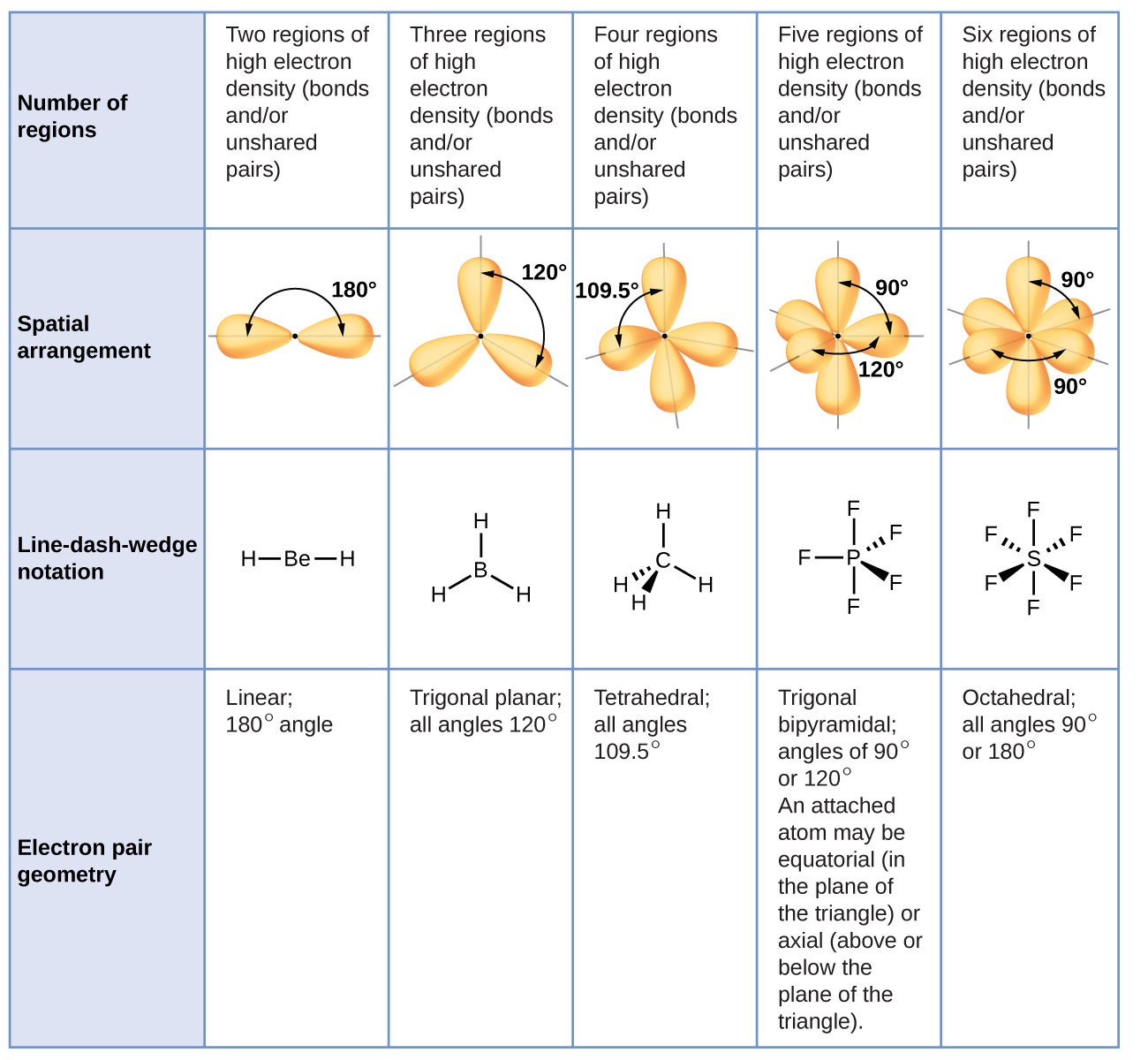
5 9 Molecular Geometry Chemistry Libretexts

Hybridization Molecular Geometry And Bond Angles Without With Lone Download Table
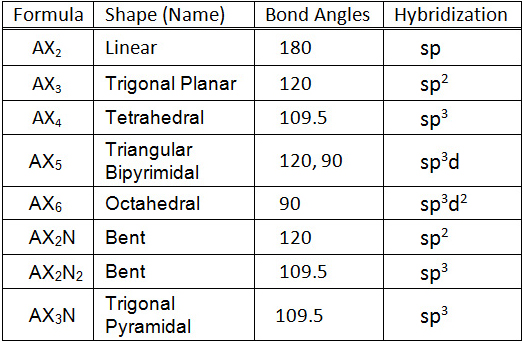
Molecular Geometry Chemical Bonding
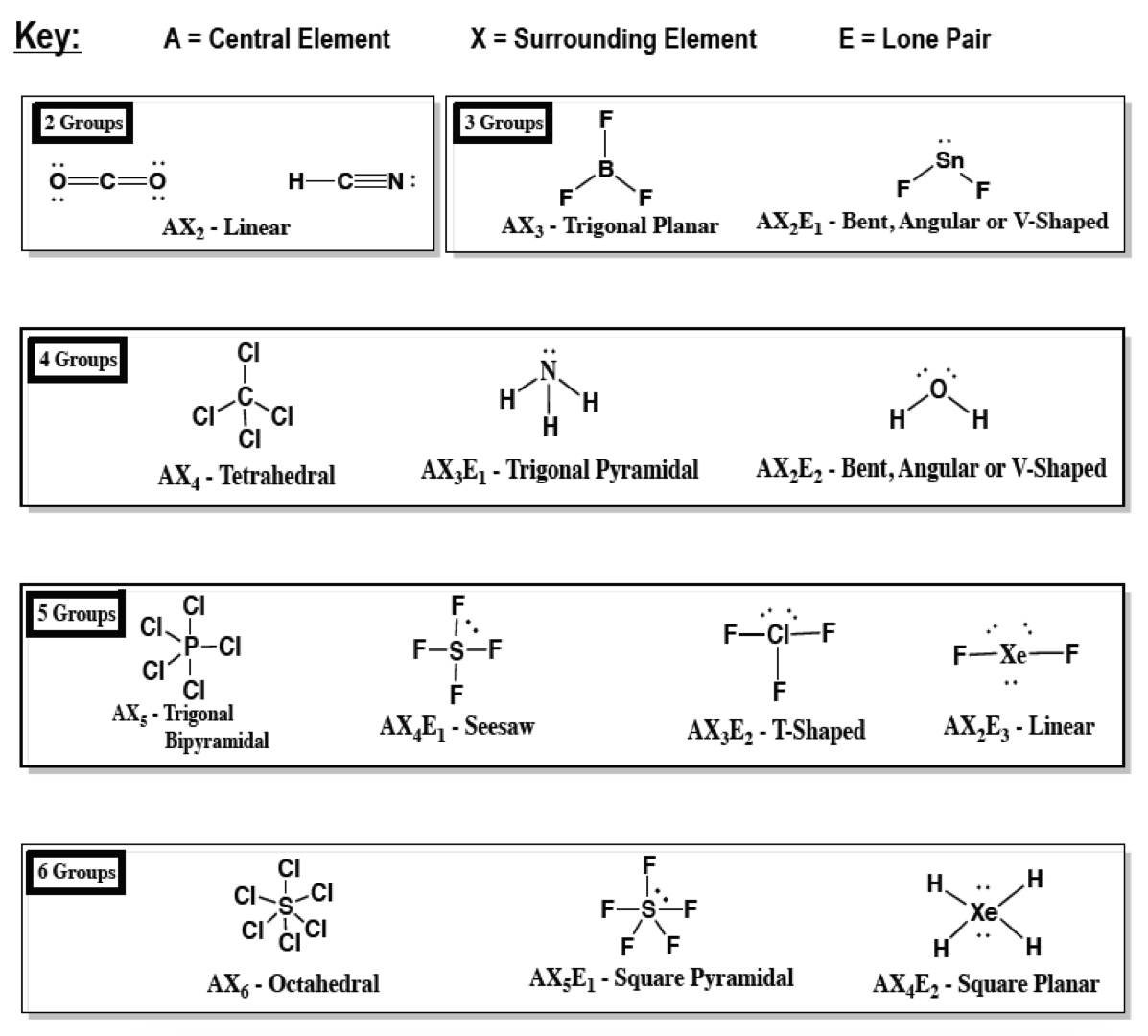
A What Is The Value Of The Bond Angles In Clutch Prep
Chem Bond Angle Scientific Tutor
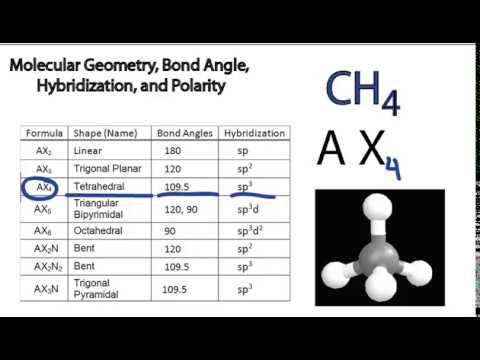
Molecular Geometry Bond Angle Hybridization And Polarity Examples Youtube

Experimental 1 And Calculated Bond Lengths Bond Angles And Dihedral Download Table
How To Predict Bond Angles Quora
Chem Bond Angle Scientific Tutor

Vsepr Theory Bond Angles Mcat Lec Youtube

Molecular Geometry Boundless Chemistry
How To Compare Bond Angles In Molecules Quora

Predicting Bond Angles Youtube

The Basic Geometry For Molecules In The Set Below Which Possesses The Greatest Bond Angle Is Which Of The Following Study Com

4 2 7 Predict The Shape And Bond Angles For Species Using Vsepr Theory Youtube